Hydrochloric acid is used in the manufacture of some explosives. Now it is a recursor, and you won’t be able to purchase it in Himmag. But it is quite easy to make at home. Chemically pure concentrated hydrochloric acid is a colorless liquid that fumes in air (hydrogen chloride is released) with a pungent odor. The density of hydrochloric acid is 1.19, at a concentration of 37%.
Reagents:
NaCl – table salt. In principle, any chloride will do, KCl for example.
H2SO4 – sulfuric acid. It must be concentrated (chars the match when cold), evaporated electrolyte is quite suitable.
Protective equipment:
Hydrochloric acid is EVIL! Causes chemical burns upon contact with skin. It shouldn't get on your clothes either - it corrodes instantly. Hydrogen chloride is poisonous! It is better to produce hydrochloric acid outdoors or in a well-ventilated area. Minimum protective equipment: 1. Rubber gloves (preferably as in the photo). 2. A gas mask or respirator (will help you quickly eliminate the consequences of a possible accident without harm to health).
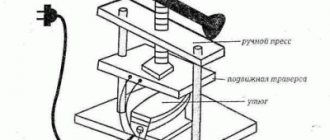
Equipment: 1. Electric stove or other heat source. 2. Libra. 3. Measuring cup. 4. Reaction vessel, I used a 250 ml flat bottom flask. 5. Safety bottle. Any flask will work perfectly (the main thing is that the volume of the flask is not less than the volume of the acid produced). 6. A pair of rubber plugs. 7. Tubes, you can use tubes from IVs.
The result should be a device like this.
True, there are a couple of nuances here: 1. The tubes used in the device must be of the same diameter; it is better to avoid using syringe needles as fittings. This will create a pressure difference and the device will explode (this happened to me when using a syringe needle). 2. It is better not to immerse the tube from which HCl comes into water, since water can be sucked into the system due to the high solubility of HCl. 3. The safety flask is, in principle, optional, you can do without it, but if water is sucked into the reaction flask and it may burst, spraying hot sulfuric acid.
Manufacturing:
1. Weigh out about 10-20 grams of salt. Take about the same amount of acid, 10-15 milliliters. I wrote approximately because the amount of acid depends on its concentration. I took 1:1 by volume. If the acid is 60-80%, then hydrogen chloride will first dissolve in water, and only then will it be released.
In short, LESS WATER – LESS PROBLEMS! So it is better to spend more time evaporating the electrolyte.
2. Place the salt in the reaction flask. 3. We assemble the installation, check all connections, they must be tight. 4. Pour 50-100 ml into a measuring cup. water (preferably distilled) and insert a tube into it. 5. We put on a gas mask. 6. Now the main thing is to quickly pour sulfuric acid into the reaction flask and plug the neck with a gas outlet tube. A gas mask will save you from the hydrogen chloride that will begin to be released. 7. At first the reaction occurs on its own, then the flask needs to be heated a little. NaCl + H2SO4 → Na2SO4 + 2HCl↑
The following are photographs of the device and the entire process:
In this way you can obtain acid with a maximum concentration of 37-40%. During the receiving process, you will see how the volume of water that you poured into the glass begins to increase. Continue saturating the water until the gas no longer dissolves. The acid should be stored in a tightly closed container to avoid volatilization of HCl. GOOD LUCK!
Currently, the choice of rechargeable batteries is huge - on sale you can find ready-to-use power sources, as well as dry-charged batteries that require preparing the electrolyte and filling it before use. Many people often carry out further battery maintenance at service centers. For various reasons, it may be necessary to prepare the solution yourself. For this event to be successful, you should know how to make electrolyte at home.
Making acid at home
Let's try to make alkali or acid at home using improvised means.
Of course, the preparation we receive will not be concentrated (this is achieved using special equipment), but the characteristic properties of the acid will certainly be noticeable. The easiest way to obtain acid at home
will be based on the electrolysis of some solution, which dissociates to form a sulfate ion. It is also possible to obtain acid in another way, but this involves either obtaining sulfur dioxide or other chemicals that may not be available, and all of them are dangerous enough to work with at home. Therefore, we obtain, for example, sulfuric acid (diluted) from copper sulfate. The concentration obtained from vitriol is not particularly dangerous, and besides, little money is needed to obtain it. So, for the experiment we need a current source (a power supply from 15 to 30 volts is perfect). We will take a graphite anode (electrode connected to the positive) so that it does not dissolve. Cathode - it is better to take the form of a graphite plate, but you can also use copper foil.
Dilute the vitriol solution and lower the electrodes into it. At the cathode we will observe the release of a brown, loose substance - this is copper.
What is copper sulfate? This is copper dissolved in sulfuric acid. Get ready to periodically remove the cathode "-" and clean it of the copper deposited on it. The longer the experiment continues, the lighter the solution of our electrolyte becomes - copper is removed from it. If we lower our indicator into a lightened solution, the color will change to scarlet. After all, sulfuric acid! Of course, it is highly diluted, but still exhibits its properties. In order to be more sure of the resulting acid, take baking soda and drop the resulting acid onto it - in this case, a rapid release of gas should be observed - this is carbon dioxide. Sulfuric acid reacts with baking soda to form sodium salt (Na2SO4), water and carbon dioxide bubbles.
It worked! For some substances it is rather weak (because it is very diluted) and you will not observe reactions with them.
Of course, you can increase the concentration of the acid by dissolving more copper sulfate in water or by evaporating the excess water in the resulting acid. I don’t recommend doing the latter, because... acid fumes are very dangerous.
- HCl - pH=1.0
- CCl3COOH - pH=1.2
- H2C2O2 - pH=1.3
- NaHSO4 - pH=1.4
- Tartaric acid – pH=2.0
- Citric acid – pH=2.1
- Lactic acid - pH=2.4
- Salicylic acid – pH=2.4
- Succinic acid – pH=2.7
- C6H5COOH – pH=2.8
- CH3COOH - pH=2.9
- NH4H2PO4 - pH=4.0
- H2S - pH=4.1
- NaH2PO4 - pH=4.5
- KH2PO4 - pH=4.7
- HCN - pH=5.1
- NH4Cl - pH=5.1
- H3BO3 - pH=5.3
- (NH4)2SO4 – pH=5.5
- Phenol - pH=5.5
- CaCO3 - pH=7.3
- (NH4)2HPO4 - pH=7.9
- C6H5COONa – pH=8.0
- NaHCO3 - pH=8.3
- CH3COONa – pH=8.9
- Na2HPO4 - pH=9.2
- Mg(OH)2 — pH=10.0
- KCN - pH=11.1
- NH3 - pH=11.3
- Na2CO3 - pH=11.6
- Na3PO4 - pH=12.0
- Ca(OH)2 - pH=12.4
- Na2SiO3 - pH=12.6
- K2S - pH=12.8
- NaOH - pH=13.0
Which acid is more acidic?!
You have probably ever asked the question “which acid is more acidic?!” “or which of the alkalis is more caustic?!” This question can be answered by considering the pH values of solutions of acids and alkalis. There are a lot of acids, so we will consider only the most basic ones. The pH value of the solution depends on the concentration. Therefore, the table shows the pH values of aqueous solutions at a concentration of 0.1 mol/l. For poorly soluble compounds marked with an asterisk, the pH of saturated solutions is indicated. The lower the pH value of the solution, the more acidic the acid, and vice versa, the higher the pH value of the solution, the more caustic the alkali! It turns out that if you drink concentrated lemon juice, the acidity of gastric juice... will decrease!? Indeed, a solution of citric acid will only dilute the stronger hydrochloric acid contained in gastric juice.
Interesting facts about sulfuric acid
Although today it is customary to produce sulfuric acid at special enterprises, it is still found in nature, and in 100% form.
For example, in Italy, on the island of Sicily, you can see H2SO4 seeping from the bottom of the Dead Sea. This place is usually called the Lake of Death; living creatures try not to come too close to it. The release of sulfuric acid from the bottom occurs due to the content of pyrite in the earth's crust. Sulfuric acid can also be released during strong volcanic eruptions. Such phenomena are quite dangerous for people and the environment. One of the consequences of H2SO4 entering the atmosphere may be climate change. Sulfuric acid is considered the main cause of acid rain, and it is released due to the release of sulfur dioxide into the air.
As previously noted, sulfuric acid effectively absorbs water from the air. This quality allows it to be used for drying gases. Previously, it was customary to pour liquid into small containers and leave it between the glasses. This helped prevent the windows in the room from fogging up.
Amount of starting substances
To prepare an electrolyte with a given density value, you need to take a strictly defined amount of starting substances. The table below shows the most common density values for different types of electrolyte.
| Density, g/cm3 | Amount of water, l | Amount of acid, l | Amount of alkali, kg | Electrolyte freezing temperature, °C |
| 1,24 | 0,819 | 0,242 | -45 | |
| 1.25 | 0,809 | 0,253 | -50 | |
| 1.26 | 0,8 | 0,263 | -55 | |
| 1.27 | 0,791 | 0,274 | -60 | |
| 1.28 | 0,781 | 0,285 | -65 | |
| 1,15 – 1.21 | 3 | 1 | -19 … +35 | |
| 1.25 – 1.27 | 2 | 1 | -20 … -40 |
To increase the permissible operating temperature in regions with hot climates or in enterprises with high ambient temperatures, caustic lithium is added to alkaline batteries in an amount of 15-20 g. per liter of electrolyte.
Electrolyte density
To fill batteries, compositions with precisely defined concentrations of constituent substances are used. To facilitate control of the quantitative composition, the concept of density was introduced. This is explained by the fact that distilled water has a density of 1 g/cm3 and any extraneous additives increase this value. Sulfuric acid and alkali have much higher specific gravity values, therefore, by measuring the density of the solution, the composition of the electrolyte can be easily determined. Density is measured using a simple device, and how to measure density, read this article .
What is formed during the interaction
The acidic residue is no longer as dangerous as the original component. It is interesting that it is the reaction with the release of carbon dioxide that soda is responsible for adding it to bread - the carbon dioxide released due to interaction with lactic acid raises the dough and makes bubbles in it.
The neutralization reaction occurs in approximately the same way: the base reacts with H2SO4 and carbon dioxide is released. Therefore, to prevent this chemical experiment from turning into a disastrous life experience, it should be carried out in a ventilated area.
On the question of how much soda is needed to neutralize H2SO4. If you take 1 part acid, 1 part alkali will be useful to react with it, that is, the ratio will be 1:1. But concentrated acid requires more soda, here the ratio will increase to 1:2. Soda solution required 3%.
In addition, if it is necessary to provide assistance for an acid burn, then prepare a solution of soda in a very approximate amount: 1 teaspoon of alkali and 2.5 glasses of water. How effective is the reaction? Feedback from those who often encounter it will help you figure this out.
Recommended for you:
How to apply and use soda ash at home
Interaction with metals
A solution of hydrochloric acid can undergo many reactions. First of all, with metals that come before hydrogen in the series of electrochemical potentials. This is the sequence in which the elements proceed as their inherent measure, the electrochemical potential (φ0), increases. This indicator is extremely important in half-reactions of cation reduction. In addition, it is this series that demonstrates the activity of metals in redox reactions.
So, interaction with them occurs with the release of hydrogen in the form of gas and the formation of salt. Here is an example of a reaction with sodium, a soft alkali metal: 2Na + 2HCl → 2NaCl +H2↑.
With other substances, interactions proceed according to similar formulas. This is what the reaction with aluminum, a light metal, looks like: 2Al + 6HCl → 2AlCl3 + 3H2↑.
Disposing of acid at home
Some acid solutions are also used in everyday life. For example, “solyanka” is used to get rid of limescale. It is also used to remove rust stains from clothing.
Popular articles Options for congratulations on March 8
Acetic acid is part of the well-known vinegar and vinegar essence.
How to dispose of expired acetic acid? To dispose of it at home, you must:
Take a plastic container with a volume twice the volume of the solution to be disposed of. To prevent the container from melting or catching fire, transfer the empty container to a bucket of ice. Pour water into an empty container Carefully pour the acid solution into the water, paying attention to the temperature of the container
Important! You cannot pour water into concentrated acid, otherwise the water may instantly boil and the acid may splash.
Using indicator paper (purchased at a specialty store), determine the pH of the solution to be disposed of. The lower the pH, the greater the amount of solution required for the neutralization reaction will be needed.
Make the solution necessary for neutralization. The substances most commonly used for this purpose (for example, magnesium hydroxide) can be purchased in stores. Carry out a neutralization reaction
The solutions prepared at the previous stage react with disposed acids, ultimately forming salts and water. The process can be considered complete if the indicator paper corresponds to a pH level of 6 - 7. Pour the resulting safe mixture down the drain (it is advisable to keep the water tap open).
Reactions with oxides
Hydrochloric acid solution also interacts well with these substances. Oxides are binary compounds of an element with oxygen that have an oxidation state of -2. All known examples are sand, water, rust, dyes, carbon dioxide.
Hydrochloric acid does not interact with all compounds, but only with metal oxides. The reaction also produces soluble salt and water. An example is the process that occurs between an acid and magnesium oxide, an alkaline earth metal: MgO + 2HCl → MgCl2 + H2O.
Reactor manufacturing attempt No. 2
I solved the problem with the reactor melting by replacing plastic with wood. I cut out a round piece from a piece of OSB and drilled 3 holes in it, two for electrodes and one for gas exhaust. I also added 2 more bolts to the design to make it easier to connect and disconnect both terminals and electrodes; the advantage was that they increased the tightness of the structure. Also, for convenience, I replaced the pipe with a shorter one and applied silicone sealant to the bottom of the wooden plug. I used the sealant as a gasket for tightness.
Bolts for easy connection
Bolts for easy connection
Bolts for easy connection
Now it seems like it shouldn’t melt, as I thought in the trailer, the idea justified itself during the test; nothing melted, however, I missed another point; after all, the lid did not fit evenly to the jar and, therefore, gas came out of the cracks since there was not sufficient tightness. I quickly tried to solve the problem with ordinary zip ties; this helped in part, but this is not a solution to the problem.
Using zip ties
The time has come to design a new reactor; let’s not give up and continue our research experiments.
CALCULATIONS WHEN PREPARING SALT SOLUTIONS
Example 1. It is necessary to prepare 500 g of a 5% solution of potassium nitrate. 100 g of such a solution contains 5 g of KN0 3;1 We make up the proportion:
100 g solution - 5 g KN0 3
500 » 1 — X
» KN0 3
5-500 „_ x= -jQg- = 25 g.
You need to take 500-25 = 475 ml of water.
Example 2.
It is necessary to prepare 500 g of a 5% CaCl solution from the salt CaCl 2 -6H 2 0. First, we perform the calculation for the anhydrous salt.
100 g solution - 5 g CaCl 2 500 "" - x
"CaCl 2 5-500 _ x = 100 = 25 g -
Molar mass of CaCl 2 = 111, molar mass of CaCl 2 - 6H 2 0 = 219*. Therefore, 219 g of CaC1 2 -6H 2 0 contain 111 g of CaC1 2. Let's make a proportion:
219 g CaC1 2 -6H 2 0-111 g CaC1 2
X
» CaС1 2 -6Н 2 0- 26 » CaCI,
219-25 x = -jjj- = 49.3 g.
The amount of water is 500-49.3=450.7 g, or 450.7 ml. Since water is measured using a measuring cylinder, tenths of a milliliter are not taken into account. Therefore, you need to measure 451 ml of water.
Chemical Spectrum Properties
Acid interacts with many metals and salts. It is considered quite strong and is on par with chamois. The main reaction manifests itself to all groups of metals located to the left of hydrogen (magnesium, iron, zinc - electrical potentials).
A diluted hydrochloric solution reacts with salts, but only with those formed by less strong acids. The well-known sodium and calcium carbonate, after interacting with it, disintegrate into water and carbon monoxide.
Nitric acid
– qualitative reaction to saline solution. To obtain it, you need to add silver nitrate to this reagent, as a result, a white precipitate will form, from which a nitrogen substance is obtained
Many interesting experiments are carried out using this mixture of water and hydrogen. For example, they dilute it with ammonia. As a result, you will get white smoke, thick, with the consistency of small crystals. Methylamine, aniline, manganese dioxide, potassium carbonate are reagents that are also susceptible to the influence of acid.
Reactions with hydroxides
This is the name given to inorganic compounds that contain a hydroxyl group –OH, in which the hydrogen and oxygen atoms are connected by a covalent bond. And, since a solution of hydrochloric acid reacts only with metal hydroxides, it is worth mentioning that some of them are called alkalis.
So the resulting reaction is called neutralization. Its result is the formation of a weakly dissociating substance (i.e. water) and salt.
An example is the reaction of a small volume of solution of hydrochloric acid and barium hydroxide, a soft alkaline earth malleable metal: Ba(OH)2 + 2HCl = BaCl2 + 2H2O.
Other uses of acid in everyday life
An acid composition can easily clean faience plumbing from limescale and rust, remove urinary stone and other contaminants. For greater effect, an inhibitor (for example, methenamine) is added to the product, which slows down the chemical reaction.
The procedure is carried out as follows: the acid is diluted with water until a 5% concentration is reached and an inhibitor is added at the rate of 0.5 g per 1 liter of liquid. The resulting composition is treated with the surface and left for 30-40 minutes (depending on the degree of contamination), after which it is washed with water.
Popular articles Stone casket
A weak acid solution is also used to remove berry stains, ink or rust from fabrics. To do this, the material is soaked in the composition for some time, after which it is thoroughly rinsed and washed as usual.
Descaling a kettle
For this purpose, use a 3-5% solution of hydrochloric acid, which is poured into a kettle and heated to 60-80°
C for 1-2 hours or until scale deposits disintegrate. After this, the scale becomes loose and can be easily removed with a wooden spatula.
The effectiveness of the method is due to the fact that the reagent reacts with magnesium and calcium carbonates and converts them into soluble salts. The carbon dioxide released during this process destroys the scale layer and makes it loose. After removing salt deposits, wash the dishes thoroughly with clean water.
Important point!
This method is not suitable for descaling enamel or aluminum kettles with chips and cracks: this will lead to corrosion of the metal and severe damage to it.
In medicine
In this area under consideration, the substance is also actively used. Dilute hydrochloric acid has the following effects:
- Digests proteins in the stomach.
- Stops the development of malignant tumors.
- Helps in the treatment of cancer.
- Normalizes acid-base balance.
- Serves as an effective remedy for the prevention of hepatitis, diabetes, psoriasis, eczema, rheumatoid arthritis, cholelithiasis, rosacea, asthma, urticaria and many other ailments.
In general, a useful drug. If a person has low acidity of gastric juice, then it will not hurt him to take a course of medications that contain hydrochloric acid. A good option is Ortho Taurine Ergo. It increases the level of hydrochloric acid in the gastric environment, helps fight bacteria and parasites.
Did you come up with the idea of diluting the acid and using it internally in this form, and not as part of medications? This is practiced, but it is strictly forbidden to do this without medical advice and instructions. By incorrectly calculating the proportions, you can swallow an excess of hydrochloric acid solution and simply burn your stomach.
By the way, you can still take medications that stimulate the production of this substance. And not only chemical ones. The same calamus, peppermint and wormwood contribute to this. You can make decoctions based on them yourself and drink them for prevention.
Making hydrochloric acid at home
Hydrochloric acid is used in the manufacture of some explosives. Now it is a recursor, and you won’t be able to purchase it in Himmag. But it is quite easy to make at home. Chemically pure concentrated hydrochloric acid is a colorless liquid that fumes in air (hydrogen chloride is released) with a pungent odor. The density of hydrochloric acid is 1.19, at a concentration of 37%.
Reagents:
NaCl – table salt. In principle, any chloride will do, KCl for example.
H2SO4 – sulfuric acid. It must be concentrated (chars the match when cold), evaporated electrolyte is quite suitable.
Protective equipment:
Hydrochloric acid is EVIL! Causes chemical burns upon contact with skin. It shouldn't get on your clothes either - it corrodes instantly. Hydrogen chloride is poisonous! It is better to produce hydrochloric acid outdoors or in a well-ventilated area. Minimum protective equipment: 1. Rubber gloves (preferably as in the photo). 2. A gas mask or respirator (will help you quickly eliminate the consequences of a possible accident without harm to health).
Equipment: 1. Electric stove or other heat source. 2. Libra. 3. Measuring cup. 4. Reaction vessel, I used a 250 ml flat bottom flask. 5. Safety bottle. Any flask will work perfectly (the main thing is that the volume of the flask is not less than the volume of the acid produced). 6. A pair of rubber stoppers. 7. Tubes, you can use tubes from IVs.
The result should be a device like this.
True, there are a couple of nuances here: 1. The tubes used in the device must be of the same diameter; it is better to avoid using syringe needles as fittings. This will create a pressure difference and the device will explode (this happened to me when using a syringe needle). 2. It is better not to immerse the tube from which HCl comes into water, since water can be sucked into the system due to the high solubility of HCl. 3. The safety flask is, in principle, optional, you can do without it, but if water is sucked into the reaction flask and it may burst, spraying hot sulfuric acid.
Manufacturing:
1. Weigh out about 10-20 grams of salt. Take about the same amount of acid, 10-15 milliliters. I wrote approximately because the amount of acid depends on its concentration. I took 1:1 by volume. If the acid is 60-80%, then hydrogen chloride will first dissolve in water, and only then will it be released.
In short, LESS WATER – LESS PROBLEMS! So it is better to spend more time evaporating the electrolyte.
The following are photographs of the device and the entire process:
In this way you can obtain acid with a maximum concentration of 37-40%. During the receiving process, you will see how the volume of water that you poured into the glass begins to increase. Continue saturating the water until the gas no longer dissolves. The acid should be stored in a tightly closed container to avoid volatilization of HCl. GOOD LUCK!
Water acidity
Most living organisms can only exist in environments close to neutral. This is due to the fact that under the influence of H+ and OH- ions, many proteins containing acidic or basic groups change their configuration and charge. And in strongly acidic and strongly alkaline environments, the peptide bond breaks, which connects individual amino acid residues into long protein chains. Because of this, ultrabasic (highly alkaline) solutions cause alkaline burns to the skin and destroy silk and wool, which are made up of protein. All living organisms are forced to maintain a certain acidity value in intracellular fluids (and since the cell consists of 80% water, then the acidity of the water
). Natural water is capable of maintaining a pH value more or less constant, even if a certain amount of acid or base enters it from the outside. If a drop of concentrated hydrochloric acid is added to a liter of distilled water, the pH will drop from 7 to 4. And if a drop of hydrochloric acid is added to a liter of river water with pH = 7, the pH will hardly change. Acids and bases entering natural water are neutralized by carbon dioxide and bicarbonate ions dissolved in it: H++HCO-3 → H2O+CO2; OH-+CO2→HCO-3.
Features of electrolyte preparation technology
When preparing yourself, remember the following:
- the density of acid and alkali is much higher than the density of water;
- reactions of mixing acid with water and dissolving alkali occur with the release of high temperatures (up to 80-90°C);
- acids and alkalis react with most metals.
From the above it follows that the dishes for preparing electrolyte must be made of material that is resistant to aggressive substances and temperature. Glassware and ceramics best meet these requirements. The use of plastic utensils is possible provided that they are not heated to high temperatures. You cannot use enamel cookware, since if there are invisible cracks in the enamel, the electrolyte will be contaminated with metal salts. The same applies to stainless steel products. Such materials do not react with water, but manufacturers do not guarantee its neutrality with respect to aggressive substances.
Preparation of acid electrolyte
The high density of the acid and the ability to heat up when mixed with water determined the specifics of preparing the solution: the acid must be poured into water. If you do the opposite, then the water on top will heat up to the boiling point and splash out along with drops of acid.
To reduce heating, it is advisable to dilute the acid in two stages. At the first stage, a solution with a density of 1.40 is prepared, and then, after cooling, the electrolyte of the required concentration is made. A solution with a density of 1.40 is called a correction solution. It is used to correct the electrolyte density in working batteries. After adding the acid to the water, the mixture is gently stirred with a glass rod. The prepared electrolyte must be left for some time (from half to a day) for its uniform mixing and complete cooling.
Preparation of alkaline electrolyte
The required amount of alkali is poured into a measured amount of water and stirred until completely dissolved. It is also necessary to wait until the sediment dissolves completely and the temperature drops to normal.
The alkali solution must be stored in a hermetically sealed container, preventing air from entering. Carbon dioxide easily reacts with alkalis to form carbonates - salts of carbonic acid. As a result, the content of the active substance in the solution decreases over time.
Solutions of acid and alkali should be transparent or have a slight yellowish tint. The presence of turbidity in the settled solution indicates low purity of the original components and are unsuitable for use in batteries.
Component Requirements
The normal course of chemical reactions places special demands on the electrolyte substances. The main requirement is a high degree of purity of materials. The purer the chemicals used to prepare the electrolyte, the higher the efficiency of the batteries and their longevity.
According to the requirements of the standards, battery sulfuric acid must contain at least 92 - 94% sulfuric acid. The remaining 6–8% is water. The content of metal salts is no more than thousandths of a percent.
Attention! The alkali is produced in dry form and has similar requirements.
If problems usually do not arise with the listed substances (responsibility for cleanliness rests with enterprises that produce materials and trade organizations), then with water things are somewhat worse. Many car enthusiasts do not distinguish between ordinary and distilled water.
Tap water is saturated with solutions of various metal salts and organic substances. Simple boiling can get rid of a small amount of hardness salts, while the remaining substances remain unchanged. In tap water, the most dangerous for batteries are iron salts, which are found there in high concentrations.
The active substances for the electrolyte must be diluted with distilled water, which differs in that its salt content is minimal. In terms of its chemical and physical parameters, such water practically corresponds to ideal water.
Use in the food industry
The food industry uses E507 in the processing of various products. Its main use in the food industry is in the production of corn syrups, especially high fructose corn syrups. It is also often found in mayonnaise and is part of citric acid, gelatin, and fructose.
Hydrochloric acid can also be used for acid modification of corn starch and for adjusting the pH of intermediates and final products.
The most common use is in the production of soft drinks, which accounts for 70-75% of demand.
E507 is also used in other areas of the food industry, including the production of hydrolyzed vegetable protein and soy sauce. It is used to acidify crushed bones, to produce gelatin, and as an acidifying agent for foods such as sauces, vegetable juices, and canned foods.
Popular articles Complex two-story flowerHydrochloric acid is often used in the production of:
- artificial sweeteners;
- lysine and choline chloride (both used primarily as animal feed additives);
- citric acid;
- corn starch;
- soft drinks;
- soy sauce.
In the field of construction
The use of hydrochloric acid and its solutions is a popular way to improve many construction processes. For example, it is often added to a concrete mixture to increase frost resistance. In addition, this way it hardens faster, and the resistance of the masonry to moisture increases.
Hydrochloric acid is also used as a limestone remover. Its 10% solution is the best way to combat dirt and marks on red brick. It is not recommended to use it to clean others. The structure of other bricks is more sensitive to the effects of this substance.
Burns and poisoning
No matter how effective this remedy is, it is dangerous. Hydrochloric acid, depending on the concentration, can cause four degrees of chemical burns:
- There is only redness and pain.
- Blisters with clear liquid and swelling appear.
- Necrosis of the upper layers of skin is formed. The blisters fill with blood or cloudy contents.
- The lesion reaches the tendons and muscles.
If the substance somehow gets into your eyes, you need to rinse them with water and then with a soda solution. But in any case, the first thing you need to do is call an ambulance.
If acid gets inside, it can cause acute pain in the chest and abdomen, swelling of the larynx, and bloody vomit. As a result - severe pathologies of the liver and kidneys.
And the first signs of vapor poisoning include a dry, frequent cough, choking, damage to teeth, burning in the mucous membranes and abdominal pain. The first emergency aid is washing and rinsing the mouth with water, as well as access to fresh air. Only a toxicologist can provide real help.
Description
A solution of hydrochloric acid belongs to the class of strong. It is colorless, transparent and caustic. Although technical hydrochloric acid has a yellowish color due to the presence of impurities of chlorine, iron and other elements. The air “smoke”.
It is worth noting that this substance is present in the body of every person. In the stomach, to be more precise, in a concentration of 0.5%. Interestingly, this amount is enough to completely destroy a razor blade. The substance will corrode it in just a week.
Unlike sulfuric acid, by the way, the mass of hydrochloric acid in solution does not exceed 38%. We can say that this indicator is a “critical” point. If you start to increase the concentration, the substance will simply evaporate, as a result of which hydrogen chloride will simply evaporate along with the water. Plus, this concentration is maintained only at 20 °C. The higher the temperature, the faster evaporation occurs.
Causes of blockages
The sewer system moves wastewater with a high content of organic matter and fats. They stick to the walls in a thin layer, which gradually grows and turns into a full-fledged blockage. As a rule, it is located near the drain hole of kitchen sinks or other plumbing fixtures. Most often, the siphon suffers from fat deposits, the bend of which actively collects sticky and viscous components of wastewater.
In addition, a common cause is foreign objects falling into the sewer. There may be hair, paper, construction waste (sand, residues of adhesive solutions), feminine pads and other unwanted components. They get stuck in bends or narrowing areas of the pipes, causing permanent blockages. No methods other than mechanical cleaning give results.
On cast iron pipes, there are cases of scale detachment from the inner surface of the walls. It remains inside and quickly becomes overgrown with fat, trapping small particles of organic matter. Mechanical cleaning of pipes will also be a solution to the problem.
Why is acid flux needed?
Metals include highly active substances. Many of them oxidize easily and quickly in the presence of air. The resulting oxides are converted into hydroxides under the influence of atmospheric moisture.
A mixture of oxidation products is clearly visible on iron products after storage in air. It's called rust. Other metals are also coated with an oxide layer, which prevents anything from being soldered to the product.
Acid fluxes help to cope with the problem, the simplest of which is soldering acid. Under this name, several different single-component or complex compositions are collected, many of which can be prepared with your own hands.
Soldering acid recipes
When carrying out soldering, acidic fluxes are often used to pretreat the surface of parts. The degree of activity of the material is selected depending on the type of metal and the degree of its contamination.
There are various products on sale, the composition of which is selected taking into account the specifics of the work to be done. You can make soldering acid at home yourself.
This will require certain knowledge, basic ability to make chemical compositions and a small amount of money to purchase components.
Why is acid flux needed?
Metals include highly active substances. Many of them oxidize easily and quickly in the presence of air. The resulting oxides are converted into hydroxides under the influence of atmospheric moisture.
A mixture of oxidation products is clearly visible on iron products after storage in air. It's called rust. Other metals are also coated with an oxide layer, which prevents anything from being soldered to the product.
Acid fluxes help to cope with the problem, the simplest of which is soldering acid. Under this name, several different single-component or complex compositions are collected, many of which can be prepared with your own hands.
What can you do at home?
The degree of acidity of fluxes differs. Active mixtures include compositions with zinc chloride. From the school course, perhaps someone remembered the properties of saline solutions.
Salts tend to undergo hydrolysis in the presence of water. Zinc chloride, when exchanged with water, forms a strong acid and a weak alkali. Therefore, the solution has an active acidic character. Hydrochloric acid quickly removes oxide substances.
Typically, soldering acid is made by adding 412 g of zinc to 1 liter of concentrated hydrochloric (hydrochloride) acid. The procedure is not entirely pleasant and safe. During operation, volatile acidic vapors are released.
You should work with concentrated hodgepodge only under a hood or in a respirator, in a well-ventilated room.
With hydrochloric acid
Several formulations with zinc chloride are popular. Zinc compounds are used in the galvanizing process to protect metals from corrosion. This chemical element is known for its resistance to oxidation processes. Making soldering acids with your own hands is not difficult.
For products made of ferrous and non-ferrous metals, the following ratio is suitable:
- zinc chloride – minimum 25%, maximum – 30%;
- concentrated hydrochloric acid – 0.7%.
Both components must be quickly dissolved in water. It should be noted that hydrochloric acid is a highly volatile substance. It is advisable to work with it under a hood. The bottle with the starting reagent should only be kept closed.
With Vaseline and alcohol
For parts made of ferrous and non-ferrous metals, sometimes it is more convenient to use a paste with acidic properties instead of soldering acid.
To prepare it, you need to mix a saturated solution of zinc chloride - 3.7% and technical petroleum jelly - 85%. To give the required consistency, add a little water to the mixture.
To work with nickel, platinum and their alloys, you can make a soldering mixture with your own hands from zinc chloride - 1.4% and ethyl (wine) alcohol - 40%. Both components must be thoroughly dissolved in water and the finished soldering solution must be mixed.
After using all the above compounds, the soldering area should be rinsed well with plain water.
With rosin
For carrying out critical work with ferrous metals, soldering precious and non-ferrous metals, a paste mixture made by yourself from rosin - 24% and zinc chloride - 1% is suitable. All this must be dissolved in ethyl alcohol. At the end of the soldering procedure, you need to wash the work area with acetone.
To form a seam with increased strength characteristics, it is recommended to take:
- rosin – 16%,
- zinc chloride – 4%,
- technical petroleum jelly – 80%.
It is more difficult to rinse the soldering area after treating it with such homemade solder paste. You need to take acetone.
Experience shows that in some cases it makes sense to replace soldering acid with a corresponding acidic paste.
Homemade sour pastes
When working with aluminum parts, a flux with oleic acid is often used, the formula C17H33COOH gives an idea of the high molecular weight. Higher acid has a viscous consistency, similar to a slightly viscous liquid.
Soldering flux is made as follows: 20 ml of oleic acid, about 3 g of lithium iodide are dissolved in a glass container in a water bath. A homogeneous solution, made with your own hands, after cooling, is poured into a glass bottle for storage.
After soldering, the working area is washed with acetone, gasoline or alcohol.
To solder nichrome, you can make a composition with your own hands from 100 g of Vaseline, 7 g of powdered zinc chloride, 7 g of glycerin.
The whole mass must be mixed well. It is advisable to do this in a thick porcelain cup or a special mortar.
Other available options
Phosphoric acid is widely used for processing iron, steel, and nickel alloys. It is often called simply phosphorus. This acid can be used to treat the surface of pure copper.
On all metals, phosphoric acid not only removes oxides, but also forms a protective layer.
During soldering work, the top layer of phosphates is easily destroyed by a heated soldering iron tip. The solder mass is evenly distributed on the processed parts. The result is a strong connection.
Perhaps the simplest option for replacing soldering acid is regular aspirin. The solution is quickly prepared with your own hands. You need to take a regular cheap tablet and dissolve it in a small amount of water. Sometimes craftsmen simply sprinkle crushed aspirin on the soldering area.
There is ready-made VTS flux on sale, which, in fact, is also soldering acid. You can make a similar product yourself.
//www.youtube.com/watch?v=Siu_1s_Hi7M
You need to take salicylic acid, Vaseline, triethanolamine. Dissolve all components in alcohol. Flux is successfully used to work with copper, platinum, silver and their alloys.
Source: //svaring.com/soldering/flus/recepty-pajalnoj-kisloty
Security measures
Preparation of electrolyte is dangerous due to the use of very aggressive substances. Concentrated solutions of acids and alkalis can cause difficult-to-heal acid burns, and if they come into contact with the eyes, they can cause blindness.
Before work, you should prepare a neutralizing solution to wash off any drops of electrolyte that accidentally fall on the body:
- 1% baking soda solution when working with acid.
- Table vinegar to neutralize alkali. The vinegar should be diluted by half with water.
You should work with rubber gloves and be sure to wear safety glasses or a mask. If electrolyte gets on your skin, you should thoroughly rinse the area of contact with a neutralizing solution, and after washing your eyes, immediately consult a doctor.
All work is carried out outdoors or in a well-ventilated area. Acid vapors released during the preparation of the solution (especially when hot) cause irritation of the upper respiratory tract, expressed by severe coughing and swelling of the mucous membranes.
As clothes at home, you can use those that you don’t really mind, since even after washing with a neutralizing solution, some of the aggressive substance will remain between the fibers of the fabric and things will be hopelessly damaged.
Transportation Features
During transit, caution should be exercised due to the highly toxic properties of the product.
It is explosive and belongs to the eighth hazard class, which includes toxic and corrosive cargo. If the carrier makes gross mistakes during transportation, it endangers not only people, but also the environment. Transportation occurs in compliance with rules that guarantee the safety of the population. It is necessary to select a stable container for transit. Tanks must be made of alloys that will not collapse under the influence of poisons. Sulfuric acid chemical tanks are suitable for transporting dangerous toxins. If it is necessary to maintain the temperature, as with a steaming liquid, thermos tanks are selected. For ordinary cargo, a sulfuric acid canister is suitable.
Transportation is permitted only in vehicles with special markings warning of dangerous cargo. Drivers who have received an ADR certificate confirming their competence are entitled to transport tanks. They are not limited in time during transportation, since they are required to comply with the speed limit to avoid the possibility of getting into an accident. To avoid situations that pose a threat to the lives of the population, the driver must drive along a specially designed route that excludes crowded places and industrial facilities.
Other types of batteries: is it possible to prepare electrolyte for them yourself?
Separately, I would like to draw attention to modern lead-acid power supplies - gel and AGM. They can also be filled with a personally prepared solution, which is in a specific form - in the form of a gel or inside separators
To refill gel batteries you will need another chemical component - silica gel, which will thicken the acid solution.
Nickel-cadmium and iron-nickel batteries
Unlike lead power sources, cadmium- and iron-nickel ones are filled with an alkaline solution, which is a mixture of distilled water and caustic potassium or sodium. Lithium hydroxide, which is part of this solution for certain temperature conditions, allows you to increase the service life of the battery.
Table 2. Composition and density of electrolyte for cadmium- and iron-nickel batteries.
It is recommended to operate nickel-iron power supplies under the same conditions as cadmium-nickel ones. However, it is worth noting that they are more susceptible to low temperatures. Therefore, they should be used up to minus 20 degrees.
Electrolyte composition
An electrolyte is a solution of the active substance in distilled water. Depending on the type of battery used, the active substance is:
- sulfuric acid for lead-acid batteries;
- alkalis (caustic sodium or potassium) for alkaline batteries.
In alkaline batteries, to meet special requirements, the electrolyte may contain the addition of caustic lithium. Also, caustic lithium is the main one in lithium-ion and lithium-polymer batteries.
Differences in electrolytes for different types of batteries
Despite the fact that the principle of operation of the solution is the same for different power sources, you should be aware of some differences in the composition. Depending on the composition, it is customary to distinguish alkaline and acidic electrolytes.
Alkaline batteries
This type of power source is characterized by the presence of nickel hydroxide, barium oxide and graphite. The electrolyte in this type of battery is a 20% solution of caustic potassium. Traditionally, the additive of lithium monohydrate is used, which allows to extend the life of the battery.
Alkaline power sources are characterized by the absence of interaction of the potassium solution with substances formed during battery operation, which helps to minimize consumption.
Acid batteries
This type of power supply is one of the most traditional, which is why the solution in them is familiar to many - a mixture of distilled water and sulfur solution. Electrolyte concentrate for lead-acid batteries is inexpensive and characterized by the ability to conduct large currents. The density of the liquid must correspond to climatic conditions.
Table 1. Recommended electrolyte density
Method for diluting electrolyte for an alkaline power source
The density and amount of electrolyte in such batteries is indicated in the operating instructions for the power source or on the manufacturer’s website.
Required solution density
The amount of solid alkali is equal to the amount of electrolyte divided by
Electrolyte is the most important component of rechargeable batteries. Without it, their work is impossible, and both the technical parameters and the durability of the batteries depend on the quality and correct preparation.
Nowadays it is possible to purchase electrolyte for all types of batteries, but sometimes there is a need to make it yourself. It is not difficult to prepare electrolyte for a battery if a number of conditions are met.
Why is analysis prescribed and how are the results deciphered?
It should be noted that a biochemical blood test to check uric acid levels is not considered standard. Doctors usually prescribe it if they suspect diseases that cause slow metabolism or kidney dysfunction.
Blood sampling is carried out in the morning on an empty stomach, usually no more than 10 ml is taken from the patient. The amount of metabolite gives the physician the opportunity to assess the condition of the organs and how correctly the body systems function.
If the content of this substance in the plasma is increased, the patient will be prescribed appropriate treatment that will prevent the accumulation of excess salts and regulate the exchange of nucleic acids.
In some laboratories, a form with the results is given directly to the patient. To decipher the result yourself, you need to know that the content of the compound is calculated in kilomoles per liter. This indicator indicates the number of molecules contained in 1 liter of blood.
About caustic and not so caustic
– These idiots placed a porcelain container with “jelly” in a special chamber, extremely isolated... That is, they thought that the chamber was extremely isolated, but when they opened the container with manipulators, the “jelly” went through the metal and plastic, like water through a blotter, and escaped outward, and everything he came into contact with again turned into “jelly.” Thirty-five people were killed, more than a hundred were maimed, and the entire laboratory building was completely unusable. Have you ever been there? Magnificent building! And now the “jelly” has flowed into the basements and lower floors... Here is the prelude to contact.
— A. Strugatsky, B. Strugatsky “Roadside Picnic”
Hello, %username%!
Blame this person for the fact that I’m still writing something. He gave me the idea.
Just after some thought, I decided that a short excursion into caustic substances would be relatively quick. Maybe someone will be interested. And for some it’s useful.
Go. Let’s immediately define the concepts.
Corrosive - 1. Chemically corrosive. 2. Sharp, causing irritation, pain. 3. Sargent, caustic.
Ozhegov S.I.
Dictionary of the Russian language. - M.: Rus.yaz., 1990. - 921 p. So, we immediately discard the last two meanings of the word. We also discard “caustic” lachrymators - which are not so much caustic as they cause lacrimation, and sternites - which cause coughing. Yes, below there will be substances that have these properties, but they are what is important! - really corrode materials, and sometimes flesh.
We will not consider substances that are caustic only for humans and the like - due to the specific destruction of cell membranes. Therefore, mustard gases will remain out of use.
We will consider compounds that are liquids at room conditions. Therefore, we will not consider liquid oxygen and nitrogen, as well as gases such as fluorine, although they can be considered caustic, yes.
As always, the view will be purely subjective, based on personal experience. And yes - it’s quite possible that I won’t remember someone - write comments, %username%, within three days from the date of publication I will supplement the article with what was forgotten from the very beginning!
And yes - I don’t have the time and energy to build a “hit parade”, so it will be a hodgepodge. And with all the exceptions, it turned out to be quite short.
Caustic alkalis
Specifically, alkali metal hydroxides: lithium, sodium, potassium, rubidium, cesium, francium, thallium (I) hydroxide and barium hydroxide. But:
- Lithium, cesium, rubidium and barium are discarded - expensive and rare
- If you, %username%, come across francium hydroxide, then the last thing you will worry about is causticity - it is terribly radioactive
- It’s the same with thallium - it’s terribly poisonous.
Therefore, sodium and potassium remain.
But let's be honest - the properties of all caustic alkalis are very similar. Sodium hydroxide is known to everyone as “caustic soda” (not to be confused with baking soda, soda ash and other soda, as well as potash). Potassium hydroxide as a food additive E525 too. Both are similar in properties: they are highly hygroscopic, that is, they attract water and “dissolve” in air. They dissolve well in water and release a large amount of heat.
“Spreading” in air is essentially the formation of very concentrated solutions of alkalis. Therefore, if you put a piece of caustic alkali on paper, leather, some metals (the same aluminum) - then after a while you will find that the material has eaten well! What was shown in “Fight Club” is very similar to the truth: indeed, sweaty hands - and the alkali - will hurt! Personally, I found it more painful than hydrochloric acid (more on that below).
However, if your hands are very dry, most likely you won’t feel anything in the dry alkali.
Caustic alkalis are excellent at breaking down fats into glycerin and salts of fatty acids - this is how soap is made (hello, “Fight Club!”) A little longer, but just as effectively, proteins are broken down - that is, in principle, alkalis dissolve flesh, especially strong solutions - and when heated . The disadvantage in comparison with the same perchloric acid (more on that below) is that all alkalis draw carbon dioxide from the atmosphere, and therefore the strength will gradually decrease. In addition, alkalis also react with the components of glass - the glass becomes cloudy, although in order to dissolve it completely, here, of course, you have to try.
Tetraalkylammonium hydroxides are sometimes classified as caustic alkalis, for example,
tetramethylammonium hydroxide
. In fact, these substances combine the properties of cationic surfactants (well, it’s like ordinary soap - only cationic: here the active diphilic particle has a “+” charge, and in soap - with a “-“ charge) and relatively high basicity. If it gets on your hands, you can lather it in water and wash it like soap; if you warm your hair, skin or nails in an aqueous solution, they will dissolve. The “causticity” against the background of sodium and potassium hydroxides is so-so.
Sulfuric acid
H2SO4
The most popular, probably, in all stories. Not the most caustic, but quite unpleasant: concentrated sulfuric acid (which is 98%) is an oily liquid that loves water very much, and therefore takes it away from everyone. By taking water away from cellulose and sugar, it chars them. In the same way, she will happily take the water away from you, %username%, especially if you pour it on the delicate skin of your face or into your eyes (well, in fact, everything will get into your eyes with adventure). Particularly kind people mix sulfuric acid with oil to make it harder to wash off and better absorbed into the skin.
By the way, when taking in water, sulfuric acid heats up greatly, which makes the picture even more juicy. Therefore, washing it off with water is a very bad idea. It’s better to use oil (rinse off, not rub in, and then rinse with water). Well, or a large flow of water to immediately cool it.
“First water, and then acid - otherwise big trouble will happen!” — this is specifically about sulfuric acid, although for some reason everyone thinks that it’s about any acid.
Being an oxidizing agent, sulfuric acid oxidizes the surface of metals to oxides. And since the interaction of oxides with acids takes place with the participation of water as a catalyst - and sulfuric acid does not release water - an effect called passivation occurs: a dense, insoluble and impenetrable film of metal oxide protects it from further dissolution.
According to this mechanism, concentrated sulfuric acid is sent to distant distances by iron and aluminum. It is noteworthy that if the acid is diluted, water appears, and it is impossible to send - the metals dissolve.
By the way, sulfur oxide SO3 dissolves in sulfuric acid and produces oleum - which is sometimes mistakenly written as H2S2O7, but this is not entirely correct. Oleum has an even greater attraction to water.
My own feelings when sulfuric acid gets on my hand: it’s a little warm, then it burns a little - I washed it off under the tap, no big deal. Don't believe the movies, but I don't recommend putting it on your face.
Organics often use chromium or “chromic mixture” - this is potassium dichromate dissolved in sulfuric acid. Essentially this is a solution of chromic acid, it is good for washing dishes from organic residues. If it gets on your hand, it also burns, but essentially it’s sulfuric acid plus toxic hexavalent chromium. You won't find holes in your hand, except maybe on your clothes.
The author of these lines knows an idiot who used potassium permanganate instead of potassium dichromate. Upon contact with organic matter, it stung a little. Those present shit themselves and escaped with a slight fright.
By the way, since we remembered chromium, let’s digress a little from the topic of acids and
Chromyl chloride
CrO2Cl2
Essentially, it is a violent compound of hexavalent chromium and hydrochloric acid. The dark red liquid, which draws water, hydrolyzes - and eventually smokes this very hydrochloric acid. Causticity is the result of this fraternal unity: chromium oxidizes, hydrochloric acid dissolves: it ignites some organic solvents (alcohol, turpentine), but dissolves in some (carbon tetrachloride, dichloromethane, carbon disulfide). It eats up metals, but not as well as acids - again it’s a matter of passivation. for example, steel, when exposed, acquires a beautiful dark blue surface.
The skin - understandably - ulcerates, and chrompica is stronger in this, since it penetrates better into the skin as into non-polar organic tissue. But it’s not even that, it’s about hexavalent chromium, which is actually a carcinogen, and therefore if it penetrates deeper, there are more problems. And of course, inhaling is much more dangerous.
Hydrochloric acid
HCl
does not exceed 38% in water. One of the most popular acids for dissolution - in this it is cooler than others, because technologically it can be very pure, and in addition to acting as an acid, it also forms complex chlorides that increase solubility. By the way, it is for this reason that insoluble silver chloride is very soluble in concentrated hydrochloric acid.
This one, when it comes into contact with the skin, burns a little more, subjectively, it also itches, and also stinks: if you work a lot with concentrated hydrochloric acid in a laboratory with a poor hood, your dentist will thank you: you’ll make it rich on fillings. By the way, chewing gum helps. But not much. Better - a hood.
Since it is not oily and does not heat up much with water, it is caustic only to metals, and not to all. By the way, steel in concentrated hydrochloric acid is passivated and says “nope!” This is what they use during transportation.
Nitric acid
HNO3
Also very popular, for some reason they are also afraid of it - but in vain. Concentrated - this is the one up to 70% - it is the most popular, higher - it is “smoking”, most often no one needs it. There is also anhydrous one - and it also explodes.
Being an oxidizing agent, it passivates many metals that become covered with an insoluble film and say “goodbye” - these are chromium, iron, aluminum, cobalt, nickel and others.
It instantly reacts with the skin according to the principle of the xanthoprotein reaction - there will be a yellow spot, which means that you, %username%, are still made of protein! After some time, the yellow skin will peel off, as if burned. At the same time, it stings less than salt, although it stinks no worse - and this time it is more toxic: flying nitrogen oxides are not very good for the body.
In chemistry, they use the so-called “nitrating mixture” - the most popular one consists of sulfuric and nitric acids. It is used in syntheses, in particular in the production of a cheerful substance - pyroxylin. In terms of causticity - the same chromium plus beautiful yellow skin.
There is also “regia vodka” - this is part nitric acid to three parts hydrochloric acid. Used to dissolve certain metals, mainly precious ones. The drip method of checking the sample of gold products is based on different ratios and the addition of water - by the way, it is very difficult for specialists using this method to fool with a fake. In terms of causticity for the skin - the same “nitrating mixture” plus it smells great, the smell cannot be confused with anything else, it is also quite toxic.
There is also “reverse aqua regia” - when the ratio is reversed, but this is a rare specificity.
By the way, about that very “smoking” one, which is red, angry and an oxidizing agent - I quote the story of a good friend who sent me just now.
I drove this very 98% nitrogen. Whether it was simply distilled for purification, or from melange, I don’t remember. I caught up with about two liters and removed the receiver. I ask the laboratory assistant to give me a clean 2-liter flask and pour it into it. She gave it to me, dry, clean, but with alcohol - and with the cap closed. That is, the pairs were and accumulated. I pour it into the funnel and pour it. I take her there and she comes back. I sprayed it well on my hands, on my face and below my neck. It felt like an eagle grabbed your face. Plus the arms, neck, under the nose, etc. little things. In my hands, I remind you, are two liters of the same goodness. Eyes closed, naturally. I understand that you can’t throw the flask, it will immediately become much worse. I carefully place the flask on the rubber stand, move to the sink, turn the gander into my face and turn on full pressure. I managed it in about five seconds. I didn’t reach the subcutaneous tissue. Otherwise everything would have been much worse. I saw with another guy that it happens after 10-15 seconds. Difficult-to-heal purple scars on half of the arm. Then I realized why she was so angry. Not only is it a fairly strong acid and oxidizing agent, it is also a wonderful solvent. It is indefinitely miscible with water, but also indefinitely miscible with, for example, dichloroethane. This is some kind of biphilic rubbish.
Phosphoric acid
H3PO4
In fact, I gave the formula for orthophosphoric acid, the most common one. And there is also metaphosphoric, polyphosphoric, ultraphosphoric - in short, that’s enough, but it doesn’t matter.
Concentrated orthophosphoric acid (85%) is such a syrup. The acid itself is average, it is often used in the food industry, by the way - when you get fillings, the surface of the tooth is first etched with phosphoric acid.
Its corrosion properties are so-so, but there is an unpleasant nuance: this syrup is well absorbed. Therefore, if it drips on things, it will be absorbed, and then it will slowly corrode. And if there is a stain or a hole from nitric and hydrochloric acid, then from phosphorus the thing will fall apart, this is especially colorful on shoes, when the hole seems to crumble until it turns out right through.
Well, in general it’s difficult to call it caustic.
Hydrofluoric acid
HF
Concentrated hydrofluoric acid is about 38%, although there are the odd exception.
A weak acid that takes the fierce love of fluoride ions to form persistent complexes with everyone with whom it can. Therefore, it surprisingly dissolves what other, stronger friends cannot, and therefore is very often used in various mixtures for dissolution. When you get it on your hand, the sensations will be greater from other components of such mixtures, but there is a nuance.
Hydrofluoric acid dissolves SiO2. That is sand. That is glass. That is, quartz. And so on. No, if you splash this acid on a window, it won’t dissolve, but a cloudy stain will remain. To dissolve, you need to hold it for a long time, or even better, heat it. When dissolved, SiF4 is released, which is so beneficial for health that it is better to do it under a hood.
A small but pleasant nuance: you, %username%, contain silicon in your nails. So, if hydrofluoric acid gets under your nails, you won’t notice anything. But you won’t be able to sleep at night - it will hurt SO much that sometimes you want to tear your finger off. Believe me, friend, I know.
And in general, hydrofluoric acid is toxic, carcinogenic, absorbed through the skin and a lot of other things - but today we’re talking about causticity, right?
Do you remember how we agreed at the very beginning that there would be no fluoride? He won't be. But they will...
Fluorides of inert gases
In fact, fluorine is a tough guy, you can’t really show off with it, and therefore some inert gases form fluorides with it. The following stable fluorides are known: KrF2, XeF2, XeF4, XeF6. All these are crystals, which in air at different speeds and readily decompose with moisture to hydrofluoric acid. The causticity is appropriate.
Hydroiodic acid
HI
The strongest (by degree of dissociation in water) binary acid. A strong reducing agent, which is used by organic chemists. In air it oxidizes and turns brown, which causes stains upon contact. The sensation upon contact is like salt water. All.
Perchloric acid
HClO4
One of the strongest (in terms of the degree of dissociation in water) acids in general (super acids compete with it - more about them below) - the Hammett acidity function (a numerical expression of the ability of a medium to be a proton donor in relation to an arbitrary base, the lower the number, the stronger the acid) is -13. Anhydrous is a strong oxidizing agent, likes to explode, and is generally unstable. Concentrated (70%-72%) is an oxidizing agent no worse, often used in the decomposition of biological objects. Decomposition is interesting and exciting because it can explode in the process: you need to make sure that there are no coal particles, that it does not boil too violently, etc. Perchloric acid is also quite dirty - it cannot be purified by subdistillation, the infection explodes! Therefore, it is not used often.
When it comes into contact with the skin, it burns and feels like salt. It stinks. When you see in films that someone threw a corpse into a container with perchloric acid and it dissolved, then yes, this is possible - but it will take a long time or warm it up. If you heat it up, it may explode (see above). So be critical of cinema (I think I saw this in 10 Cloverfield Lane).
By the way, the causticity of chlorine oxide (VII) Cl2O7 and chlorine oxide (VI) Cl2O6 is the result of the fact that these oxides form perchloric acid with water.
Now let’s imagine that we decided to combine strong acidity and the causticity of fluorine in one compound: let’s take a molecule of perchloric or sulfuric acid and replace all the hydroxyl groups on it with fluorine! The rubbish will turn out to be rare: it will interact with water and similar compounds - and at the site of the reaction a strong acid and hydrofluoric acid will immediately be obtained. A?
Fluorides of sulfur, bromine and iodine
Remember we agreed to consider only liquids? For this reason, our article did not include chlorine trifluoride ClF3
, which boils at +12 °C, although all the horror stories are that it is terribly toxic, ignites glass, a gas mask and, when spilled 900 kilograms, eats 30 cm of concrete and a meter of gravel - everything This is true. But we agreed - liquids.
However, there is a yellow liquid - iodine pentafluoride IF5
, colorless liquid -
bromine trifluoride BrF3
, light yellow -
bromine pentafluoride BrF5
, which are no worse. BrF5, for example, also dissolves glass, metals and concrete.
Similarly, among all sulfur fluorides, only disulfur decafluoride (sometimes called sulfur pentafluoride) is liquid - a colorless liquid with the formula S2F10
. But this compound is quite stable at ordinary temperatures, does not decompose with water - and therefore is not particularly caustic. True, it is 4 times more toxic than phosgene with a similar mechanism of action.
By the way, iodine pentafluoride is said to have been the “special gas” used to fill the atmosphere in the escape shuttle in the final scenes of the 1979 film Alien. Well, I don’t remember, honestly. Reminds me! Damn, it’s so cool there that I couldn’t resist and dedicated a separate article to it.
I even found it, took a closer look and realized that Ripley lived there in such harsh conditions that the alien beast was just cute
Super acids
The term "super acid" was coined by James Conant in 1927 to classify acids that are stronger than ordinary mineral acids.
In some sources, perchloric acid is classified as a super acid, although this is not so - it is an ordinary mineral one. A number of superacids are mineral acids to which a halogen has been attached: the halogen pulls electrons onto itself, all the atoms get very angry, and everything goes to hydrogen as usual: it falls off in the form of H+ - boom: so the acid has become stronger.
Examples - fluorosulfuric and chlorosulfuric acids
Fluorosulfuric acid has a Hammett function of -15.1; by the way, thanks to fluorine, this acid gradually dissolves the test tube in which it is stored.
Then someone smart thought: let's take a Lewis acid (a substance that can accept a pair of electrons from another substance) and mix it with a Brønsted acid (a substance that can donate a proton)! Antimony pentafluoride was mixed with hydrofluoric acid to obtain hexafluorantimony acid HSbF6
. In this system, hydrofluoric acid releases a proton (H+), and the conjugate base (F−) is isolated by a coordination bond with antimony pentafluoride. This produces a large octahedral anion (SbF6−), which is a very weak nucleophile and a very weak base. Having become “free”, the proton determines the hyperacidity of the system - Hammett function -28!
And then others came and said why they took Bernstead’s weak acid and came up with this.
Trifluoromethanesulfonic acid
- in itself is already a super acid (Hammett function -14.1).
So, they added antimony pentafluoride to it again - they got a decrease to -16.8! The same trick with fluorosulfuric acid gave a reduction to -23. And then a group of scientists from the chemistry department of the American University of California, led by Professor Christopher Reed, hung out with colleagues from the Institute of Catalysis of the Siberian Branch of the Russian Academy of Sciences (Novosibirsk) and came up with carborane acid H(CHB11Cl11). Well, they called it “carborane” for ordinary people, but if you want to feel like a scientist, say “2,3,4,5,6,7,8,9,10,11,12-undecachlor-1-carba-closo-dodecaborane (12)” three times and quickly.
This is what this beauty looks like
This is a dry powder that is soluble in water.
This is the Strongest Acid at the moment. Carborane acid is approximately a million times stronger than concentrated sulfuric acid. It is not possible to measure the strength of an acid on a conventional scale, since the acid protonates all known weak bases and all solvents in which it dissolves, including water, benzene, fullerene-60, and sulfur dioxide. Subsequently, Christopher Reed told Nature news service: “The idea for the synthesis of carborane acid was born out of fantasies about “molecules that had never been created before.” Together with his colleagues, he wants to use carborane acid to oxidize atoms of the noble gas xenon - simply because no one has done it before. Original, what can I say.
Well, since super acids are ordinary acids, they act normally, only a little stronger. It is clear that the skin will burn, but this does not mean that it will dissolve. Fluorosulfonic acid is a separate case, but it’s all thanks to fluorine, just like in fluoride.
Trihaloacetic acids
Specifically, trifluoroacetic and trichloroacetic acid
Cute and pleasant due to the combination of the properties of an organic polar solvent and a fairly strong acid.
They stink—like vinegar. The cutest thing is trifluoroacetic acid: a 20% solution destroys metals, cork, rubber, bakelite, polyethylene. The skin burns and forms dry ulcers that reach the muscle layer.
Trichloroacetic acid is the younger brother in this regard, but that’s okay too. By the way, applause to the weaker sex: in pursuit of beauty, some go for the so-called TCA peeling procedure (TCA is TetraChloroAcetate) - when this same trichloroacetic acid is used to dissolve the top, rough layer of skin.
True, if a cosmetologist chats on the phone, a failure is possible
Acetic acid
CH3COOH
Most likely, you have this acid in your kitchen - and yes, it is used as a food additive E260. But it can also be stronger - a 70-80% aqueous solution of acetic acid is called acetic essence, and if the concentration is close to 100% - glacial acetic acid (because it can freeze and form something similar to ice.
Acetic acid is not as caustic towards metals as mineral acids, but since it is not so polar, and to some extent even diphilic (a combination of hydrophobic and hydrophilic parts in one molecule - as in surfactants), it is great absorbed by the skin. Solutions with acetic acid concentrations greater than 30% are considered dangerous. The peculiarity of burns is that the development of coagulation necrosis of adjacent tissues of varying length and depth is also initiated - if not washed off, there will be long-healing ulcers and scars.
Well, it stinks, of course, quite noticeably.
Formic acid
HCOOH
We have already discussed that formic acid, which is formed in the body after taking methanol, is one of the main reasons for its toxicity. So, formic acid from the outside is not at all so dangerous, since it is quickly metabolized and eliminated by the body. Toxicity is quite low - for rats LD50 is about 1.8 g / kg, and therefore formic acid is also often used, including as a food additive - and there is no need to be afraid of this.
The “causticity” of formic acid depends on the concentration. According to the classification of the European Union, a concentration of up to 10% has an irritating effect, and more than 10% has a corrosive effect. And again we are not talking about metals and glass - but about the body. Upon contact with skin, 100% liquid formic acid causes severe chemical burns. Contact of even a small amount of it on the skin causes severe pain; the affected area first turns white, as if covered with frost, then becomes wax-like, with a red border appearing around it. The acid easily penetrates the fatty layer of the skin, so washing the affected area with a soda solution must be done immediately. So the ants really do know something.
Bromine
Br2
A heavy, caustic liquid of red-brown color with a strong unpleasant odor, vaguely reminiscent of the smell of both iodine and chlorine. By the way, the name “bromine” comes from the Greek βρῶμος - “stinker”, “smelly”.
Bromine is a typical halogen; in terms of chemical activity, bromine occupies an intermediate position between chlorine and iodine. That is, not as fast as fluorine - but livelier than boring iodine. And yes, it doesn’t reach chlorine either.
Slightly soluble in water, well soluble in some organic solvents. Bromine water, a reagent for unsaturated hydrocarbons, stinks, but is quite peaceful and does not dissolve anything much.
Pure bromine is powerful, smelly and hairy, as well as toxic. When it comes into contact with the skin, it causes burns: the trouble is that bromine molecules are non-polar, and therefore penetrate well into hydrophobic human skin and flesh - and therefore burns are really painful, take a long time to heal, and almost always leave a scar. Aluminum flares up on contact with bromine, other metals are more temperate, but in powder form - some react, for example, iron.
Concrete and glass are quite resistant to bromine. Organic compounds with bromine - what? - Right! - are brominated in the presence of an unsaturated bond. For this reason, the stability of polymers depends on their type, for example polyethylene and polypropylene - they didn’t care about bromine at room conditions.
Hydrogen peroxide
H2O2
An unstable compound that constantly breaks down gradually into oxygen and water. The higher the concentration, the more unstable it is, which gradually turns into an explosive hazard. To stabilize technical hydrogen peroxide, sodium pyrophosphate or sodium stannate is added to it; When stored in aluminum containers, a corrosion inhibitor is used - ammonium nitrate.
Hydrogen peroxide in the laboratory is usually a 38% solution. Upon contact with skin, it causes a chemical burn with a characteristic white color. The burn is painful, especially on thin skin; the whitened, keratinized skin then often cracks and itches.
In medicine, 3% hydrogen peroxide is used to clean deep wounds with a complex profile, purulent streaks, phlegmons and other purulent wounds, the sanitation of which is difficult - the substance not only has an antiseptic effect, but also creates a large amount of foam when interacting with the enzyme catalase. This, in turn, makes it possible to soften and separate necrotic areas, blood clots, and pus from the tissues, which will be easily washed away by subsequent injection of an antiseptic solution into the wound cavity. By the way, hydrogen peroxide is undesirable in other cases of wounds: while having good cleansing properties, this substance does not actually speed up the healing process, since it damages the cells adjacent to the wound, as well as young, newly formed tissues - and this is also fraught with the formation of scars.
Except for burns on the skin, it does not corrode or dissolve anything. Metals, glass and plastics are resistant to hydrogen peroxide.
Hydrogen peroxide also gave the world many unique natural blondes with black hair roots!
Close to hydrogen peroxide are the so-called peracids - acids that contain peroxide groups. Example: peracetic acid CH3COOOH
- a substance that resembles hydrogen peroxide in its properties, and therefore is used in exactly the same areas.
There is “pervomur” or “S-4” (no, this is not the C-4 you thought about) - this is performic acid HCOOOH
, which is even weaker than peracetic, and therefore surgeons wash their hands with it before surgery.
And finally - trifluoroperacetic acid CF3COOOH
- a fierce, crazy oxidizing agent, which organic chemists look at with admiration for the possibility of oxidizing aniline to nitrobenzene, obtaining hypervalent iodine in organic compounds, the Bayer-Villiger reaction and other things that are poorly understood by normal people. In terms of causticity - trifluoroacetic acid mixed with hydrogen peroxide, which, in fact, is what it is, and therefore poses a particular danger to the hands, yes. Due to its high oxidizing power, trifluoroperacetic acid is not sold, but is usually obtained by admiring organic chemists directly, where necessary, by reacting trifluoroacetic anhydride with hydrogen peroxide.
Well, something like this, if we talk about liquid and causticity. Will there be more additions?